How Water Balance Makes for Safer Stay-at-Home Swimming
I started this series on pool water and health before any of us knew a coronavirus was about to upset the whole world. I’ve long held that a diligent owner can be more certain about a pool’s health at home than at many public facilities. Now we can add social distancing to the advantages a home pool offers.
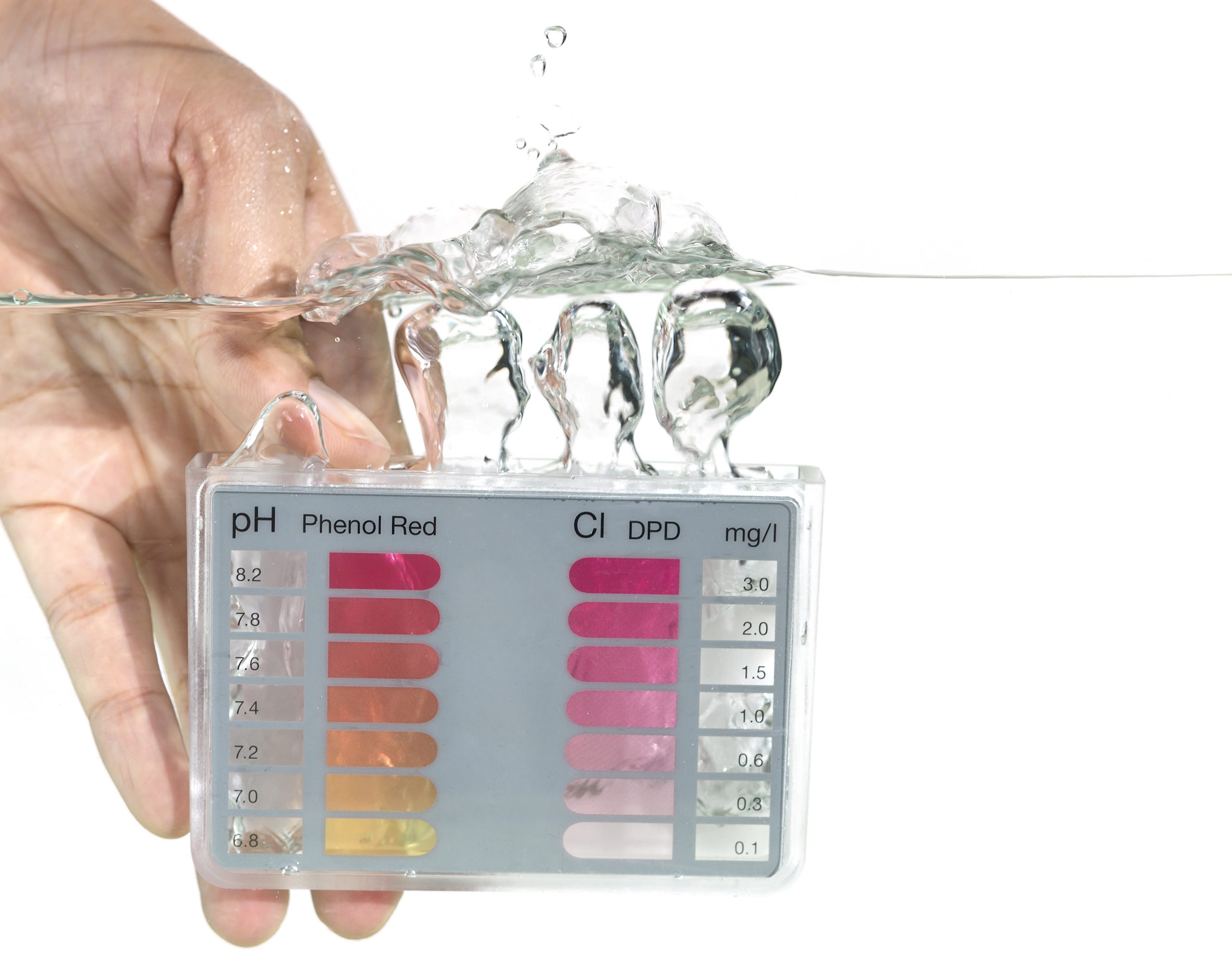
If you own a pool, you already know that chlorine kills viruses in the water. But you need to know more about your water’s chemistry to be sure the chlorine is doing its job. That’s where water balance comes into the picture. When water isn’t balanced, there isn’t enough free chlorine to kill the virus. At that point adding more chlorine doesn’t help and could further compound your problems.
Pool chemistry is more complicated than many people realize. Let’s look at the five factors you need to measure and test before taking action:
- pH level of pool or spa water
- Total Alkalinity
- Calcium level
- Dissolved Solids level
- Temperature
Let’s look at how these five factors impact one another and the health of your pool water.
pH level
Your pool’s pH (potential Hydrogen) level has to be kept balanced between 7.4 (the level maintained by the human body) and 7.6. Water pH is balanced at 7.0. Under that it’s acidic; over, it’s alkaline.
When pH is out of range, bad things happen faster, doing damage to people and pools. It can even affect how well the chlorine protects you against microbes. Out of balance pH leads to other chemical reactions. For example when the pH is too high (alkaline) chlorine combines with hydrogen to make hydrochloric acid. Adding more chlorine in this situation just makes more acid.
Total alkalinity
Total alkalinity is a measure of pH stability. Use this measure to help you keep pH levels from fluctuating too much. Test and adjust alkalinity first, then test and adjust pH.
Total Calcium Hardness
Total Calcium Hardness indicates if water is hard or soft. You want pool water just a little soft, about 150 ppm. When water is too soft, it doesn’t have enough calcium and will leach out calcium from other sources in contact with the water. A favorite target is the calcium in plaster pool walls. Too much calcium makes pool water too hard, freeing excess calcium to migrate to other pool surfaces. You don’t want to discover nasty white scales on your pool walls or clogging pipes. That’s a sure sign of hard water shedding excess calcium.
Total Dissolved Solids (TDS)
Total Dissolved Solids (TDS) is a measure of organic matter, minerals, metals, and salts dissolved in your pool water. A high TDS level leads to pool stains, algae blooms, and scaling. It also depletes the chlorine available to do its job of keeping water healthy.
Temperature
Temperature affects how chemicals act and react with one another in your pool water. High temperatures speed up chemical reactions. Rising temperatures lead to rising pH levels (more alkaline) and dropping chlorine levels. Temperature also affects water test results, which is why it’s important to get your water sample to the testing facility without delay.
Getting Calcium Right with LSI
In the next installment, we’ll look at how these same five factors figure in the Langelier Saturation Index (LSI). Dr. Langelier’s Index helps keep calcium balanced in pool water.